The science of washing cloth diapers
Most cloth diaper wash advice tells you what to do. This page tells you why — the actual chemistry of how surfactants surround urine residue, how detergent enzymes break down proteins and oils, why hard-water minerals neutralize cleaning power, and what's mechanically happening inside the drum during agitation. If you've ever wanted to understand the physics underneath the wash routine recommendations, this is the page.

The short version: cloth diapers get clean because surfactants surround oily soils into spherical structures called micelles, enzymes break down proteins and fats into water-soluble fragments, and agitation knocks both kinds of soil loose from deep inside absorbent fibers — then the rinse cycle physically carries everything away in the wash water. When a wash routine fails, it's almost always because one of those processes was too weak to do its job.
What cloth diapers actually contain
A used cloth diaper isn't just "dirty." It contains four chemically distinct categories of soil, and each requires a different cleaning mechanism to remove.
| Soil type | What it is chemically | What removes it |
|---|---|---|
| Urine salts | Urea, sodium, potassium, chloride — water-soluble compounds | Water and rinsing alone, when fibers are accessible |
| Body oils & sebum | Lipids — long-chain fatty acid molecules, hydrophobic | Surfactant micelles + lipase enzymes |
| Bacteria & their byproducts | Living and dead microorganisms, plus their metabolic waste (including ammonia) | Surfactants lift them off; rinse carries them away. Heat or chemical sanitizers kill survivors. |
| Organic residue (skin cells, fecal matter) | Mixed protein, fat, and complex carbohydrate — chemically heterogeneous | Protease + lipase + amylase enzymes, working in combination |
This is why "use more detergent" is sometimes the right answer and sometimes not — depending on which type of soil is the actual problem. Underdosing affects all four. Hard water disproportionately affects the surfactant-driven removal of oils and bacteria. Cold water disproportionately affects enzyme activity, which matters most for protein and starch residues.
How surfactants work — the molecular mechanism
A surfactant molecule has two ends with opposite preferences. One end — the hydrophilic head — is attracted to water. The other end — the hydrophobic tail — avoids water and is attracted to oils and fats. This dual-personality structure is what makes detergent work.
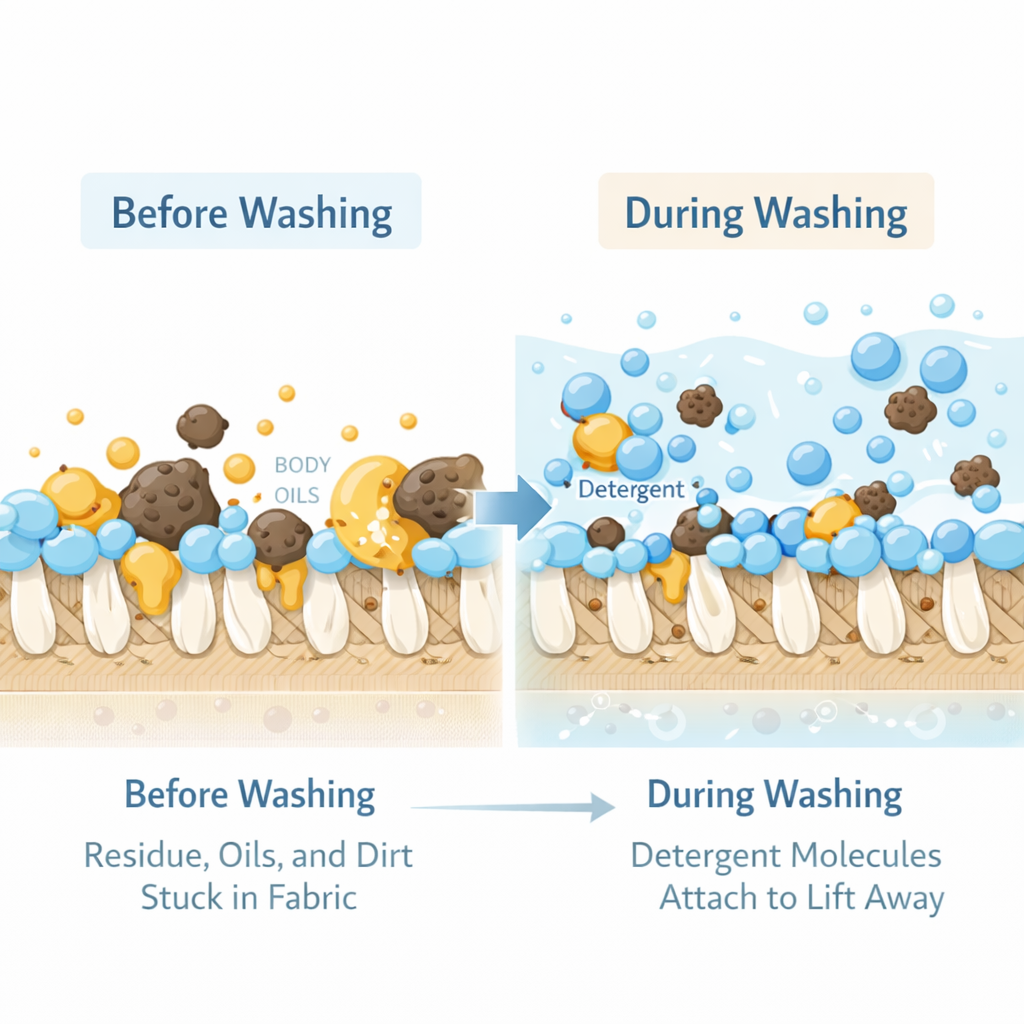
Here's what happens when you add detergent to a wash:
- Surface tension drops. Surfactant molecules collect at the water's surface, with their hydrophilic heads in the water and their hydrophobic tails in the air. This breaks the cohesive bonds between water molecules at the surface and lets water actually wet the fabric instead of beading on it. Without this step, water can't even reach the soils trapped inside the absorbent core.
- Tails attach to oils. Inside the fabric, the hydrophobic tails of free surfactant molecules find the oily soils — sebum, body fats, urine lipids — and embed themselves in them.
- Micelles form. Once enough surfactant molecules are surrounding an oil droplet (the threshold is called the critical micelle concentration), they organize into a sphere. Tails point inward, surrounding the oil. Heads point outward, presenting a water-friendly surface to the surrounding wash water.
- The micelle floats free. The encapsulated oil is now wrapped in a water-soluble package. It detaches from the fabric and stays suspended in the wash water.
- The rinse carries it away. The rinse cycle replaces the dirty wash water with clean water, taking the suspended micelles with it.
This is why detergent dose matters so much. Below the critical micelle concentration, micelles don't form properly — the surfactant molecules just float around individually without organizing. There's enough to drop surface tension but not enough to actually package up and remove oils. Underdosing is essentially asking surfactants to do the third step of the process without enough molecules present to form the structures that do the work.
Anionic surfactants (negatively charged heads) are the most common in laundry detergent — powerful but vulnerable to hard-water ions. Non-ionic surfactants (no charge) are weaker per molecule but unaffected by hard water. Most laundry detergents combine both: anionics for cleaning power, non-ionics for hard-water tolerance. This is why hard-water-friendly detergents work in mineral-rich water where pure anionic formulas don't.
Enzymes — the targeted cleaners
Surfactants handle oils and physical soil suspension. Enzymes handle the rest — the protein, starch, and complex molecules that surfactants alone can't dissolve. Modern laundry detergents typically contain four to six enzyme types, each engineered to break down a specific category of molecule.
Enzymes are catalysts — they participate in the chemical reaction that breaks down soils, then return to their original state and start again. A small amount of enzyme can process a lot of substrate, which is why detergents only need to contain a few percent enzyme by weight to be highly effective.
Why temperature affects enzymes specifically
Enzymes are proteins with three-dimensional shapes that determine which molecules they bind and break apart. They have an optimal temperature range (typically 30–50°C / 86–120°F for most laundry enzymes) where this shape is most active. Above their optimum, enzymes denature — the protein unfolds and stops working. Below their optimum, they're slower but still functional.
Modern laundry detergents are engineered with cold-active enzyme variants, but most still work better in warm water than cold. This is part of why warm water cleans cloth diapers better than cold — not just because oils dissolve more easily, but because the enzymes themselves operate closer to peak efficiency.
It's also why the 130°F (54°C) ceiling for cloth diaper safety is conveniently close to the upper end of enzyme optimum — you're getting full enzyme activity without damaging PUL or elastics. Sanitary cycles at 150–160°F not only damage diapers; they also begin to denature the very enzymes you're paying for in your detergent.
Why warm water helps — the thermodynamics
Beyond enzyme activity, water temperature affects cleaning through three physical mechanisms.
- Oil viscosity drops with heat. Body oils and lipid soils flow more easily at warmer temperatures. Surfactants can penetrate them more readily and form micelles faster. Below about 70°F, many lipids stiffen enough to resist surfactant action.
- Molecular motion increases. Higher temperature means molecules move faster. Surfactant molecules collide with soils more often, water molecules carry suspended micelles away from fibers more efficiently, and dissolved soil compounds diffuse out of the fabric more quickly.
- Solubility of polar compounds rises. Urea, urine salts, and other water-soluble residues dissolve more readily in warm water. The same fabric that holds these compounds tightly in cold water releases them more freely when warmed.
There's a temperature sweet spot for cloth diapers: roughly 90–120°F (32–49°C). This range gives enzymes full activity, dissolves lipids well, keeps molecular motion high, and stays well below the threshold where PUL waterproof layers and elastics begin to degrade. Higher temperatures don't add cleaning benefit; they just add wear on the diapers.
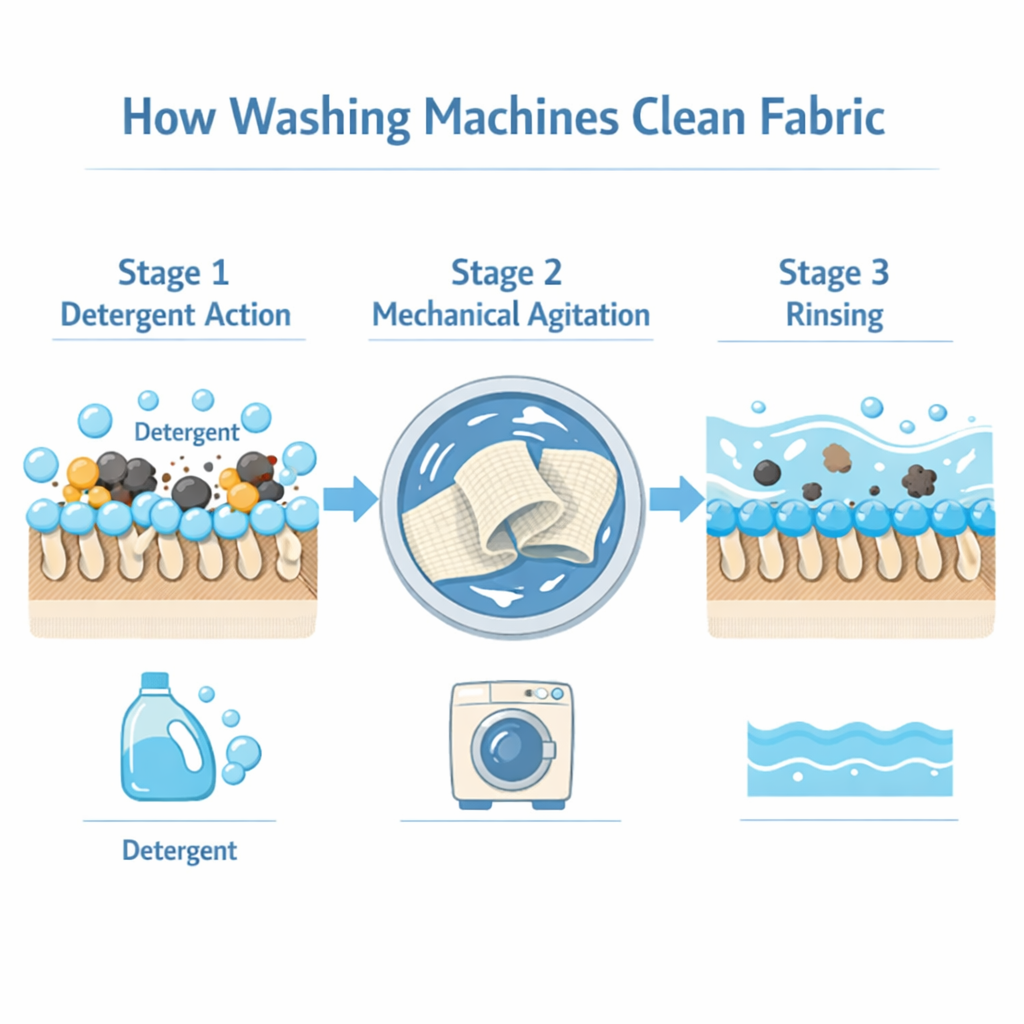
Agitation — the mechanical side of cleaning
Detergent chemistry can lift soils off fabric only after the chemistry has reached the soils. In an absorbent material like cloth diaper fabric, that's a non-trivial physical problem.
Cloth diapers contain layers of hemp, cotton, or bamboo fibers. Soil — urine, oils, bacteria — gets distributed deep within those layers when the diaper is in use. To clean the fabric, wash water carrying surfactants and enzymes has to physically penetrate to where the soils live, react with them, and then carry the now-loosened residue back out.
Three mechanical effects drive this:
- Bulk water flow. Wash water moves through the fabric, carrying surfactants in and soils out. The faster the flow, the better the exchange.
- Fiber-on-fiber friction. When fabric rubs against other fabric in the drum, the mechanical force loosens soil from individual fibers, exposing it to the surrounding water and detergent. This is why agitation matters more than people realize.
- Compression and release. Drum tumbling repeatedly compresses and releases the absorbent material. Each cycle squeezes contaminated water out of the fibers and lets clean wash water flow back in — a kind of automated rinsing happening continuously during the wash.
Why drum loading matters so much
For all three mechanical effects to work, fabric needs to actually rub against other fabric and get squeezed. This requires a properly loaded drum:
- Underloaded drum: diapers swim freely without contacting each other. No fiber-on-fiber friction. Water flow happens, but the loosening effect of mechanical contact is missing. Cleaning suffers significantly.
- Properly loaded (2/3 to 3/4 full): diapers regularly contact each other during tumbling. Friction and compression-release cycles operate normally. Best cleaning conditions.
- Overloaded drum: diapers can't tumble freely — they pack together, and movement of individual items relative to each other is reduced. Compression-release barely happens. Wash water can't penetrate as easily. Cleaning suffers.
This is why "wash more diapers" is sometimes the right answer when smell or absorbency problems appear. It's not that the detergent is wrong; it's that the mechanical part of the cleaning process isn't running.
Why hard water reduces cleaning power
Hard water is water containing dissolved calcium and magnesium ions. The U.S. Geological Survey classifies water as soft below 60 ppm of dissolved minerals, moderately hard 60–120 ppm, hard 120–180 ppm, and very hard above 180 ppm. Most U.S. tap water falls in the moderately hard to hard range.
The problem for cloth diaper washing is that calcium and magnesium ions interact directly with anionic surfactant molecules. The negative charge on the surfactant head attracts the positive charge on the calcium ion (Ca²⁺), and the two bond into an insoluble compound. The surfactant is now neutralized — it can't form micelles, can't lift oils, can't do its job.
Worse, the calcium-surfactant compound doesn't just sit in solution. It precipitates onto fabric, forming the gritty residue that older cleaning literature calls "soap scum." On cloth diapers, this residue:
- Coats fibers and reduces absorbency by blocking the fiber's ability to wet
- Traps bacteria in the residue layer, where they survive the wash and produce ammonia later
- Holds organic soil against the fabric instead of letting it rinse away
This is why the right answer to hard water isn't more detergent — it's a water softener. Adding more detergent just produces more calcium-surfactant compound, which makes the residue problem worse. A water softener (Calgon, Borax, or similar) contains compounds called chelating agents that bind to calcium and magnesium ions before the surfactant does. With those ions tied up, the surfactant remains free to do its actual job.
A whole-house water softener swaps calcium and magnesium ions for sodium ions (which don't react with surfactants) before the water ever reaches the washing machine. Households with whole-house softeners can typically use about 25% less detergent than they would with hard water and still get equivalent cleaning — because every detergent molecule is now actually available to clean. If you've ever wondered why the same detergent works perfectly for one family and barely cleans for another, water hardness is usually the answer.
The rinse cycle — where soil actually leaves
Here's a fact that surprises most people: the wash cycle doesn't actually remove soil from cloth diapers. It only loosens it. The rinse cycle is what physically takes the soil out of the machine.
During the wash cycle:
- Surfactants form micelles around oils
- Enzymes break down proteins, fats, and starches into smaller fragments
- Agitation knocks soil loose from fibers
- Soil ends up suspended in the wash water — lifted off the fabric, but still inside the drum
If the wash cycle ended without rinsing, all that suspended soil would settle back onto the fabric as the water drained, redepositing it on the now-clean fabric. This is why the rinse step isn't optional or decorative — it's the only mechanism by which soil physically leaves the diaper.
For the rinse to work properly:
- The wash water has to be replaced fully. A short or weak rinse leaves dirty water (and suspended soil) in the drum.
- The rinse needs enough volume. HE machines use less water by design, which sometimes means an extra rinse is needed for heavily-soiled cloth diapers.
- Suds need to be cleared. Visible suds in the final rinse mean the rinse hasn't fully completed — surfactants are still in the fabric, and so is whatever they were carrying.
If you're seeing suds at the end of a wash, an extra rinse cycle is the right response. It's also why washing diapers with the rest of the household laundry is risky — the additional fabric volume can absorb wash water and make rinsing less complete.
Why two cycles work better than one long one
A common question is why cloth diaper wash routines use a short pre-wash followed by a long main wash, rather than just one really long wash cycle. The answer comes down to soil concentration in the wash water.
During the pre-wash, surfactants and water lift the bulk of the surface soil off the diapers. By the end, the wash water contains a high concentration of suspended soil — urine, oils, bacteria, organic residue. If the same water then continued into the main wash, that suspended soil would interfere with the deep-cleaning step. There's a chemistry term for this: surfactants have a finite carrying capacity for suspended soil, and a saturated solution can't pick up much more.
By draining the pre-wash water and starting the main wash with clean water and fresh detergent:
- The diapers enter the main wash already partially clean
- The fresh detergent has its full surfactant capacity available
- Clean water can carry away soils from deeper inside the absorbent layers
- The final rinse leaves diapers in genuinely clean water rather than diluted dirty water
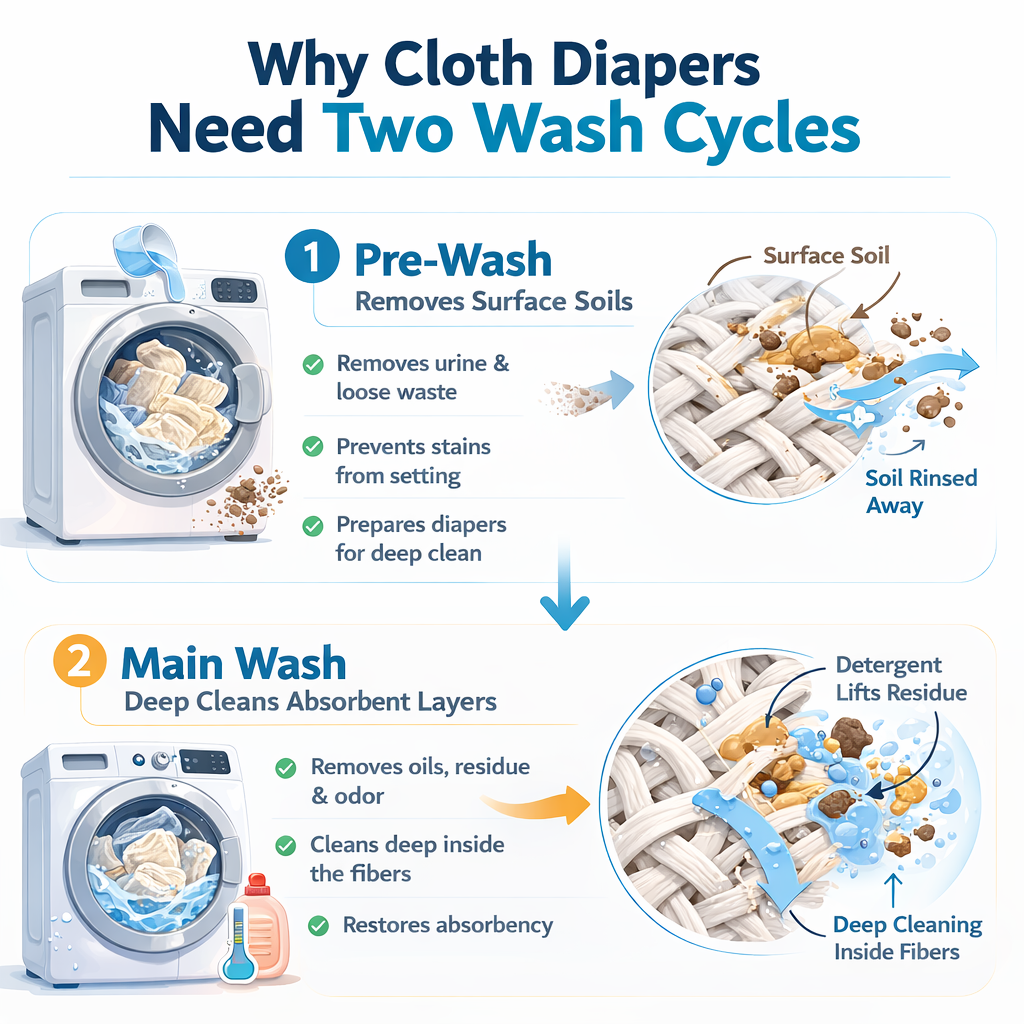
It's the same principle as washing dishes by hand. Rinsing a plate under running water before scrubbing with soap is more effective than scrubbing a plate covered in stuck food — the soap can do its actual job because the easy soil is already gone.
Common questions
How does laundry detergent actually clean cloth diapers?
What are the enzymes in laundry detergent and what do they do?
Why does temperature matter so much for cloth diaper washing?
Why doesn't more detergent fix hard water problems?
Does the wash cycle actually clean the diapers, or does the rinse?
Why does drum loading matter for cleaning?
What is the critical micelle concentration and why does it matter?
Why do cloth diapers smell when wet but not dry, scientifically?
Is the chemistry the same for adult cloth diapers as for baby cloth diapers?
Putting the science into practice
Questions about the chemistry of your specific situation — particular detergent, water hardness, machine type? Contact us and we'll work through the science with you.
